Accreditations
ISO/IEC 17025 Sampling Organization Accreditation
What is ISO/IEC 17025 Sampling Organization Accreditation?
ANAB sampling organization accreditation program is designed for organizations that specialize in the collection of samples used for various testing and inspection activities.
The ANAB sampling organization accreditation program will help assure the quality of sample collection used for testing and inspection by assuring compliance to the international standard ISO/IEC 17025 and ANAB accreditation requirements based on national and international requirements.
Accredited sampling organizations are required to operate under a quality management system and have processes and procedures in place for assuring the quality of the environmental sample collection activities performed. This includes requirements for the competency of staff, validity in the sampling methodology and techniques used, and correct performance of the sample collection activities.
Our goal through accreditation is to improve the quality of the organizations performing sample collection activities and the quality of the samples collected. Accreditation of sampling organizations can support all industries relying on high quality sample collection to assure quality of their data.
ANAB accreditation managers and sampling experts have practical experience in the field and are often viewed as peers to the sampling organizations. The ANAB sampling organization accreditation program is well-established and should be your choice for accreditation as an organization that performs sample collection activities.
Steps to ISO/IEC 17025 Sampling Organization Accreditation
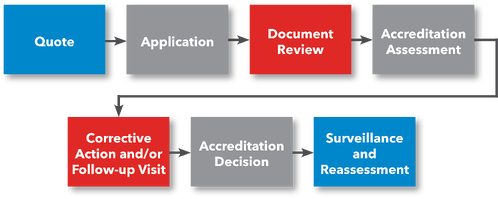
Steps for Getting Accredited
- Request a Quote
- File Application
- Prepare for Accreditation Assessment
- Submit Documentation for Review
- (Optional) Preliminary Assessment
- Accreditation Assessment
- Corrective Action (if applicable)
- ANAB Accreditation Decision
- Receive Accreditation Certificate
ISO/IEC 17025
General Requirements for the Competence of Testing and Calibration Laboratories
ISO/IEC 17025 specifies the general requirements for the competence, impartiality and consistent operation of laboratories.
ISO/IEC 17025 is applicable to all organizations performing laboratory activities, regardless of the number of personnel.
Laboratory customers, regulatory authorities, organizations and schemes using peer-assessment, accreditation bodies, and others use ISO/IEC 17025 in confirming or recognizing the competence of laboratories.
Talk to an Expert
Patrick Selig
Senior Manager of Accreditation, Inspection, Laboratories, and Related Activities
414-501-5475

Need Training To Support Your Accreditation Journey?
Learn at your own pace with online courses or choose an instructor led class offered online or in a convenient location.

